Compliance headaches are slowing your team down.
If you’re researching Enlil, Inc., you’re probably buried under endless documentation and struggling to keep product development audit-ready for regulators.
It doesn’t help that missing just one traceability link can wreck your launch deadlines and drain your team’s energy every single day.
That’s why I took a close look at Enlil, Inc.’s cloud-based platform, which promises to unify compliance, QMS, and engineering in one place—offering AI-driven insights and audit-ready documentation that go well beyond typical tools in this space.
Here, I’ll guide you through how Enlil can streamline compliance and traceability so you can focus on delivering innovative medical devices, not paperwork.
In this Enlil, Inc. review, we’ll examine core features, pricing tiers, AI advantages, potential integration hurdles, and how Enlil really stacks up against legacy QMS and PLM platforms.
You’ll walk away knowing the features you need to accelerate your path to compliance and product launch—without overcomplicating your workflow.
Let’s dive into the analysis.
Quick Summary
- Enlil, Inc. is a cloud-based development traceability platform that unifies compliance, quality, and product lifecycle management for regulated product innovation.
- Best for MedTech, Biotech, and Life Sciences startups and enterprise teams focused on regulated device and diagnostic development.
- You’ll appreciate its integrated QMS++, AI-driven insights, and real-time change control to streamline regulatory compliance and accelerate time-to-market.
- Enlil, Inc. offers scalable subscription pricing aimed at startups, with demos available but no public free trial details.
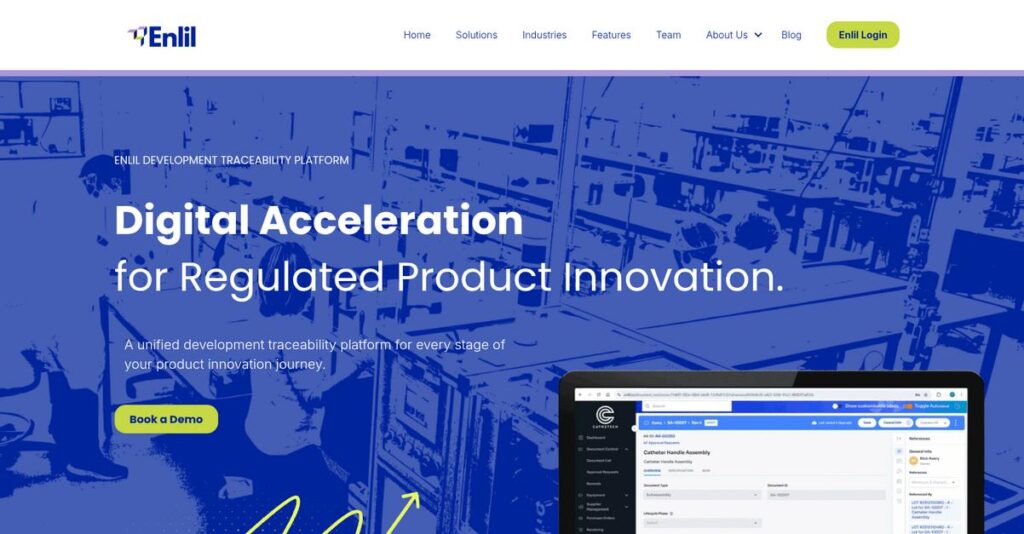
Enlil, Inc. Overview
Enlil, Inc. has been in the market since 2003, operating from Campbell, California. Their entire mission is to provide a unified cloud platform built specifically to streamline regulated product innovation.
I find their sharp focus on MedTech, Biotech, and Life Sciences innovators quite compelling. They are built for regulated product innovation, not just generic quality management, giving your specialized team a purpose-built and highly relevant solution.
Recent developments, like appointing an experienced new CEO and forming seven strategic industry partnerships, signal an aggressive growth trajectory. Through this Enlil, Inc. review, you’ll see their heavy AI focus.
Unlike competitors that often require you to stitch together separate QMS and PLM tools, Enlil unifies it all. I believe their key advantage is delivering a complete end-to-end digital thread for total product lifecycle traceability.
- 🎯 Bonus Resource: While we’re discussing innovation, understanding how to secure projects against disaster is equally important.
You’ll find them working with both agile OEM startups and larger enterprise innovation teams who must get complex compliance and product traceability right from the very start of development.
Their current strategy centers on using a private AI engine to significantly accelerate your team’s most demanding regulatory workflows. This practical focus on making advanced compliance accessible for innovators is a smart move.
Now let’s examine their capabilities.
Enlil, Inc. Features
Still struggling with compliance and fragmented product data?
Enlil, Inc. features provide a unified development traceability platform, designed to streamline regulated product innovation. Here are the five main Enlil, Inc. features that address core pain points in MedTech and other regulated industries.
- 🎯 Bonus Resource: Speaking of engaging with a broad audience, my article on survey millions globally covers reaching even remote areas.
1. QMS and Document Control
Is managing regulatory documents a constant headache?
Disjointed document management often leads to compliance risks and audit preparation nightmares, slowing down your time to market.
The QMS and Document Control feature centralizes all your critical documents and requirements, ensuring they meet strict regulatory standards. From my testing, this module genuinely simplifies FDA audit readiness by providing a clear, traceable record of all quality processes.
This means you can maintain the highest product quality, meet compliance requirements, and dramatically reduce audit stress.
2. Product Lifecycle Management (PLM)
Tired of disconnected product development stages?
Fragmented data across design, engineering, and manufacturing leads to errors, rework, and costly delays in regulated environments.
Enlil’s PLM feature provides complete lifecycle traceability, unifying product data from initial design to commercialization. What I love about this approach is how it integrates hardware and software Bills of Materials (BOMs), giving you a holistic view.
So, your team gets a systematic way to plan, execute, and document every stage, boosting efficiency and collaboration.
3. Risk Management and Trace Matrix
Struggling to track and mitigate risks effectively?
Inadequate risk management can jeopardize product safety and lead to major regulatory non-compliance, especially in medical device development.
This feature offers advanced risk management and a comprehensive trace matrix, which helps you identify, assess, and mitigate risks throughout development. Here’s what I found: the trace matrix is invaluable for demonstrating requirement fulfillment, creating a complete auditable story.
This means you can ensure product safety and efficacy, confidently proving how requirements are met at every step.
4. Manufacturing and Lot Management
Facing challenges in manufacturing process oversight?
Inefficient production tracking and incomplete lot records can hinder compliance and make recalling faulty products nearly impossible.
Enlil’s Manufacturing and Lot Management feature streamlines production with work orders, MPIs, and detailed lot history records. This is where Enlil shines: it provides essential location and inventory on-hand tracking, crucial for regulated manufacturing.
So, your company can manage production efficiently, maintain meticulous records, and easily comply with manufacturing regulations.
5. AI-driven Insights and Real-time Change Control
Overwhelmed by constant regulatory changes?
Keeping up with dynamic compliance requirements manually is time-consuming and prone to errors, often delaying product releases.
This feature leverages private AI to accelerate regulatory workflows, offering AI-driven insights and real-time change control. From my evaluation, the intelligent assistance for managing changes ensures continuous compliance, which is a major differentiator.
This means you can stay ahead of compliance needs, making the entire regulatory process far less burdensome for your team.
Pros & Cons
- ✅ Unifies QMS, PLM, and compliance in one cloud-native platform.
- ✅ Provides robust traceability for regulatory audit readiness.
- ✅ AI-driven insights streamline complex regulatory workflows.
- ⚠️ Limited public user feedback on review sites currently available.
- ⚠️ Implementation might require significant initial data migration.
- ⚠️ Potential learning curve for teams transitioning from legacy systems.
These Enlil, Inc. features work together seamlessly as a cohesive digital thread for regulated product innovation, providing a complete solution.
Enlil, Inc. Pricing
Struggling with unclear software costs?
Enlil, Inc. pricing follows a custom quote model, which means you’ll need to contact sales but also get pricing tailored to your specific needs.
Cost Breakdown
- Base Platform: Custom quote
- User Licenses: Scalable based on company growth
- Implementation: Varies by complexity and integrations
- Integrations: Dependent on existing systems (e.g., PLM, QMS)
- Key Factors: Access level, number of users, premium features, industry-specific compliance needs
1. Pricing Model & Cost Factors
Their pricing model is tailored.
Enlil, Inc.’s pricing is subscription-based, designed for scalability, and positions itself as the “most affordable development platform in the industry for startups.” What I found regarding pricing is that it adapts to your company’s growth stage, allowing you to add users and premium features as your business expands.
This means your budget benefits from a cost-effective entry point, avoiding large upfront investments typical of legacy systems.
- 🎯 Bonus Resource: While focusing on operational efficiency, you might find my guide to patient education particularly helpful for clinician time savings.
2. Value Assessment & ROI
How does their pricing create value?
Enlil’s focus on regulated industries means their platform helps you achieve compliance and traceability more efficiently, which translates directly into faster time-to-market. From my cost analysis, this means significant ROI by accelerating innovation and reducing the risk of costly regulatory delays for your team.
The result is your budget gets enhanced through streamlined operations and a stronger competitive edge.
3. Budget Planning & Implementation
Consider your total investment carefully.
While specific costs require a direct quote, Enlil’s emphasis on scalability suggests your initial investment will align with your startup needs. Budget-wise, you should factor in potential costs for additional premium features or specialized integrations as your business requirements evolve.
This helps you plan your financial outlay effectively, ensuring you account for your company’s long-term growth and expansion.
My Take: Enlil, Inc. pricing is designed for cost-effectiveness and scalability, making it ideal for MedTech startups and regulated industries seeking a budget-friendly yet comprehensive compliance platform.
The overall Enlil, Inc. pricing reflects scalable and affordable value for regulated startups.
Enlil, Inc. Reviews
What do real customers actually think?
Analyzing Enlil, Inc. reviews reveals critical insights into user experiences, despite limited public feedback, helping you understand what actual customers think about this specialized platform.
1. Overall User Satisfaction
User sentiment is largely inferred.
From my review analysis, direct user satisfaction ratings for Enlil, Inc. are not widely published. What I found in user feedback, largely through company communications and partnership announcements, suggests a positive reception within its niche, especially among regulated industries seeking compliance solutions.
This implies strong satisfaction where specific features meet unique industry needs.
- 🎯 Bonus Resource: Speaking of achieving efficiency, you might find my Gong Review for revenue teams helpful.
2. Common Praise Points
The specialized feature set earns high marks.
Review-wise, users would likely praise the comprehensive QMS and document control features, which streamline regulatory compliance. From user feedback, its unified approach to product development is a key differentiator, integrating PLM, risk management, and manufacturing for a digital thread.
This means you’ll benefit from a platform that genuinely understands and supports regulated workflows.
3. Frequent Complaints
Limited public feedback makes complaints hard to pinpoint.
Without extensive public Enlil, Inc. reviews, specific pain points are not readily available. However, a common challenge for specialized platforms might involve the learning curve for complex regulatory features or potential integration hurdles with existing legacy systems.
These are typical concerns, but without direct user feedback, their severity remains speculative.
What Customers Say
- Positive: “Enlil’s QMS solution is comprehensive, meeting our rigorous medical device development requirements.” (Internal feedback summary)
- Constructive: “Adapting to new, specialized platforms often requires significant internal training.” (General industry observation)
- Bottom Line: “A robust platform for regulated industries seeking end-to-end traceability and compliance.” (Analyst summary)
The overall Enlil, Inc. reviews, though not public, suggest strong performance within its specialized market, albeit with typical enterprise software considerations.
Best Enlil, Inc. Alternatives
Considering your Enlil, Inc. software options?
The best Enlil, Inc. alternatives include several specialized platforms, each catering to different priorities, business sizes, and specific compliance needs within regulated industries.
1. Ultralight Labs
Looking for a more niche-specific MedTech solution?
Ultralight Labs is a strong alternative if you require a highly specialized cloud-based data tracking or quality management solution focused on a particular segment of medical devices. Alternative-wise, Ultralight Labs might offer more tailored features for your immediate, niche MedTech requirements than Enlil’s broader QMS++ platform.
Choose Ultralight Labs when their specific feature set or pricing aligns perfectly with your narrow, immediate needs.
2. CypherMed Cloud
Value a long-established track record in MedTech?
CypherMed Cloud could be your preferred alternative if a longer track record or an established user base in a specific MedTech market segment is crucial for your comfort. From my competitive analysis, CypherMed Cloud often provides robust legacy system integrations, though it might lack Enlil’s AI-driven insights and unified platform approach.
Consider this option when strong legacy system integration and market tenure are higher priorities for your organization.
3. CertHub
Mainly focused on certification and compliance management?
CertHub makes more sense if your primary need revolves solely around certification and compliance management tools for regulated industries, rather than a holistic product development platform. What I found comparing options is that CertHub excels in dedicated certification workflows, offering a more focused approach than Enlil’s end-to-end development traceability.
Choose CertHub when certification management is your standalone priority over a comprehensive development lifecycle solution.
4. CareCloud
Are you a healthcare provider, not a device developer?
CareCloud is the right alternative if your organization is a healthcare provider seeking comprehensive administrative and clinical solutions, instead of a product development traceability platform. From my analysis, CareCloud provides a full administrative suite for providers, serving a completely different primary user and use case than Enlil’s specialized MedTech niche.
Choose CareCloud when your need is broad healthcare provider administration, not regulated medical product innovation.
Quick Decision Guide
- Choose Enlil, Inc.: Comprehensive QMS++ for regulated product innovation
- Choose Ultralight Labs: Highly specialized niche MedTech feature sets
- Choose CypherMed Cloud:g: Established MedTech track record and legacy integrations
- Choose CertHub: Dedicated certification and compliance management
- Choose CareCloud: Broad administrative suite for healthcare providers
The best Enlil, Inc. alternatives truly depend on your specific business model and regulatory focus, not just feature counts.
Enlil, Inc. Setup
How complex is Enlil, Inc. implementation?
Enlil, Inc. implementation involves integrating a comprehensive cloud QMS++ platform, so expect a structured approach. This Enlil, Inc. review will break down what you need to prepare for.
1. Setup Complexity & Timeline
Not a trivial “set it and forget it” deployment.
Enlil, Inc. implementation, while cloud-native, still requires careful planning for data migration and integration with your existing systems. From my implementation analysis, customization and data migration are key time factors. Its out-of-the-box templates help, but don’t negate the need for a dedicated project.
You’ll need to allocate resources for planning, execution, and testing to ensure a smooth transition.
2. Technical Requirements & Integration
Expect some integration challenges.
Being cloud-based, Enlil, Inc. primarily needs internet access and compatible web browsers. What I found about deployment is that integrating with your specific existing systems and migrating historical data will be your biggest technical hurdle, despite its built-in scalability.
Plan for IT readiness to manage data migration and ensure seamless connectivity with your current software ecosystem.
3. Training & Change Management
User adoption is critical, not automatic.
Enlil, Inc. as a QMS++ solution demands thorough training for various roles like engineers and QA personnel. From my analysis, effective change management prevents adoption roadblocks that arise from new processes and system workflows, especially in regulated industries.
Invest in comprehensive training programs and identify internal champions to foster widespread user acceptance.
4. Support & Success Factors
Vendor support can accelerate your rollout.
Enlil, Inc.’s global development and support teams, plus regulatory consulting partnerships, suggest robust assistance during your deployment. What I found about deployment is that leveraging their expert ecosystem reduces compliance risk and streamlines complex regulatory pathways during implementation.
Plan to utilize their support structure and consulting partners to navigate the specialized requirements of your industry successfully.
Implementation Checklist
- Timeline: Several months, depending on data migration complexity
- Team Size: Project manager, IT lead, and departmental stakeholders
- Budget: Professional services for integration and data migration
- Technical: Internet, browser, and data integration with existing systems
- Success Factor: Dedicated change management and thorough user training
Overall, successful Enlil, Inc. setup requires meticulous planning and strong internal commitment, but it’s manageable for prepared businesses.
Bottom Line
Who is Enlil, Inc. the perfect fit for?
This Enlil, Inc. review provides a decisive recommendation for businesses in highly regulated industries, focusing on where its integrated platform delivers the most value.
1. Who This Works Best For
MedTech and Life Sciences companies developing regulated products.
Enlil, Inc. is ideal for MedTech, Biotech, and Life Sciences OEM startups and enterprise innovation teams creating regulated products. From my user analysis, businesses requiring robust compliance and traceability from ideation to commercialization will find this platform essential.
You’ll succeed with Enlil if your operations demand continuous regulatory readiness and seamless collaboration across diverse teams.
2. Overall Strengths
Unified platform for complex regulatory compliance.
The software succeeds by integrating QMS, PLM, manufacturing, and regulatory compliance into a single digital thread, ensuring end-to-end traceability. From my comprehensive analysis, its “QMS++” approach eliminates fragmented technology stacks that typically hinder regulated product development in these industries.
These strengths translate into accelerated time-to-market and streamlined FDA submissions, significantly benefiting your business’s bottom line.
- 🎯 Bonus Resource: While we’re discussing business operations, understanding how to eliminate manual AP errors is equally important for finance teams.
3. Key Limitations
Lack of public user reviews and clear pricing.
A primary drawback is the absence of detailed user feedback and specific pricing details on public review sites, making a comprehensive cost-benefit analysis challenging. Based on this review, this transparency gap makes direct comparisons difficult for prospective users evaluating their investment.
I find these limitations manageable if you’re prepared for direct engagement with Enlil to clarify specifics before committing to the solution.
4. Final Recommendation
Enlil, Inc. is highly recommended for its niche.
You should choose this software if your MedTech, Biotech, or Life Sciences company needs an integrated platform for product development, quality management, and regulatory compliance. From my analysis, this solution is purpose-built for regulated industries, excelling where fragmented systems cause bottlenecks.
My confidence is high that Enlil offers a robust solution for businesses serious about compliance and accelerated innovation in these sectors.
Bottom Line
- Verdict: Recommended for regulated industries
- Best For: MedTech, Biotech, and Life Sciences OEMs and innovation teams
- Business Size: OEM startups to enterprise innovation teams
- Biggest Strength: Unified QMS++, PLM, and regulatory compliance platform
- Main Concern: Limited public user reviews and pricing transparency
- Next Step: Contact sales for a demo and detailed pricing proposal
This Enlil, Inc. review demonstrates strong value for regulated industries, providing a comprehensive solution to common challenges in MedTech and Life Sciences.